|
The five parts can be arranged to form a clock-like location for the electrons in any atom. Once the atomic number is
known, each electron has an address into the atom which can be read off in order around the clock.
| The Cube has a space for every electron-address |

|
|
|
 |
|
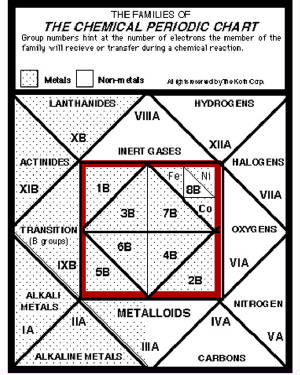
The 2-D representation of the cube make writing and note taking easy as lessons are discussed. Here we see the names
of the Chemical Families on the Periodic Chart ordered and organized graphically.
| The student notebook leaflet |
|
|
|

